Clinical Evaluation of OH-0101 (Withamax) in Breast Cancer Patients Receiving Chemotherapy
Background
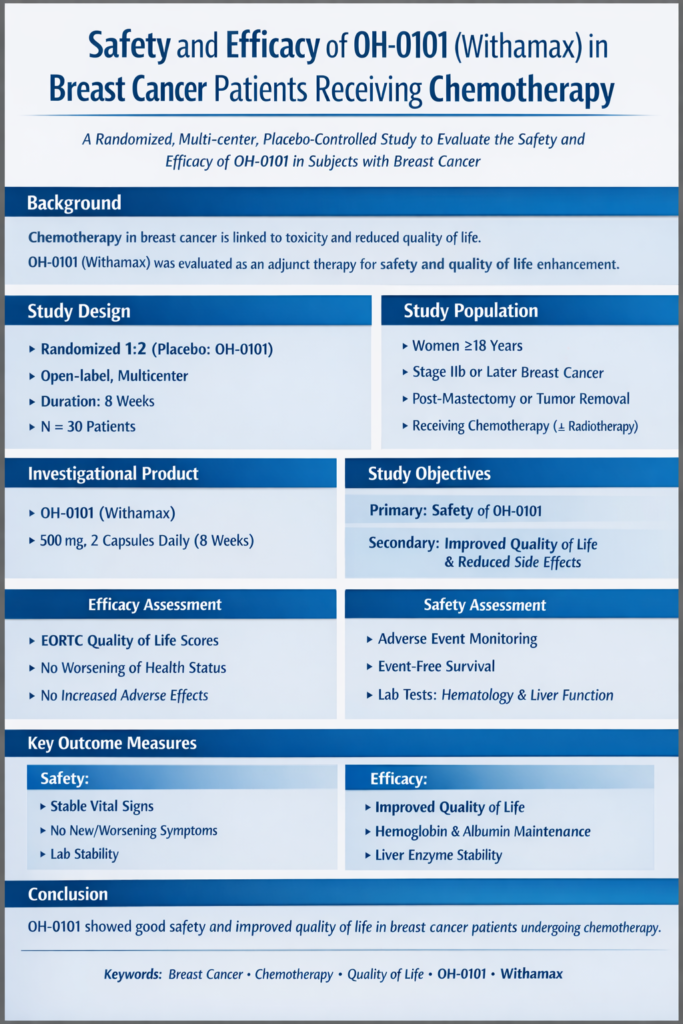
Chemotherapy in breast cancer is frequently associated with systemic toxicity, hepatic stress, hematological alterations, and deterioration in patient-reported quality of life. Supportive care strategies that improve treatment tolerance without compromising safety remain clinically relevant. The REFORM I Ca study was conducted as a proof-of-concept randomized evaluation to assess the safety and adjunct efficacy of OH-0101, a concentrated extract of Withania somnifera, administered during chemotherapy.

Thirty female subjects aged 18 years and above with Stage IIb or later breast cancer were enrolled following mastectomy or tumor removal. All participants were receiving chemotherapy, with optional radiotherapy. Subjects were randomized in a 2:1 ratio, with 20 assigned to the investigational arm and 10 to control. OH-0101 was administered at a dose of 500 mg twice daily for eight weeks.
Methods
The study was designed as a randomized, placebo-controlled, open-label, multicenter clinical evaluation. Assessments were conducted at screening, baseline, 14 days, 28 days, and 56 days.
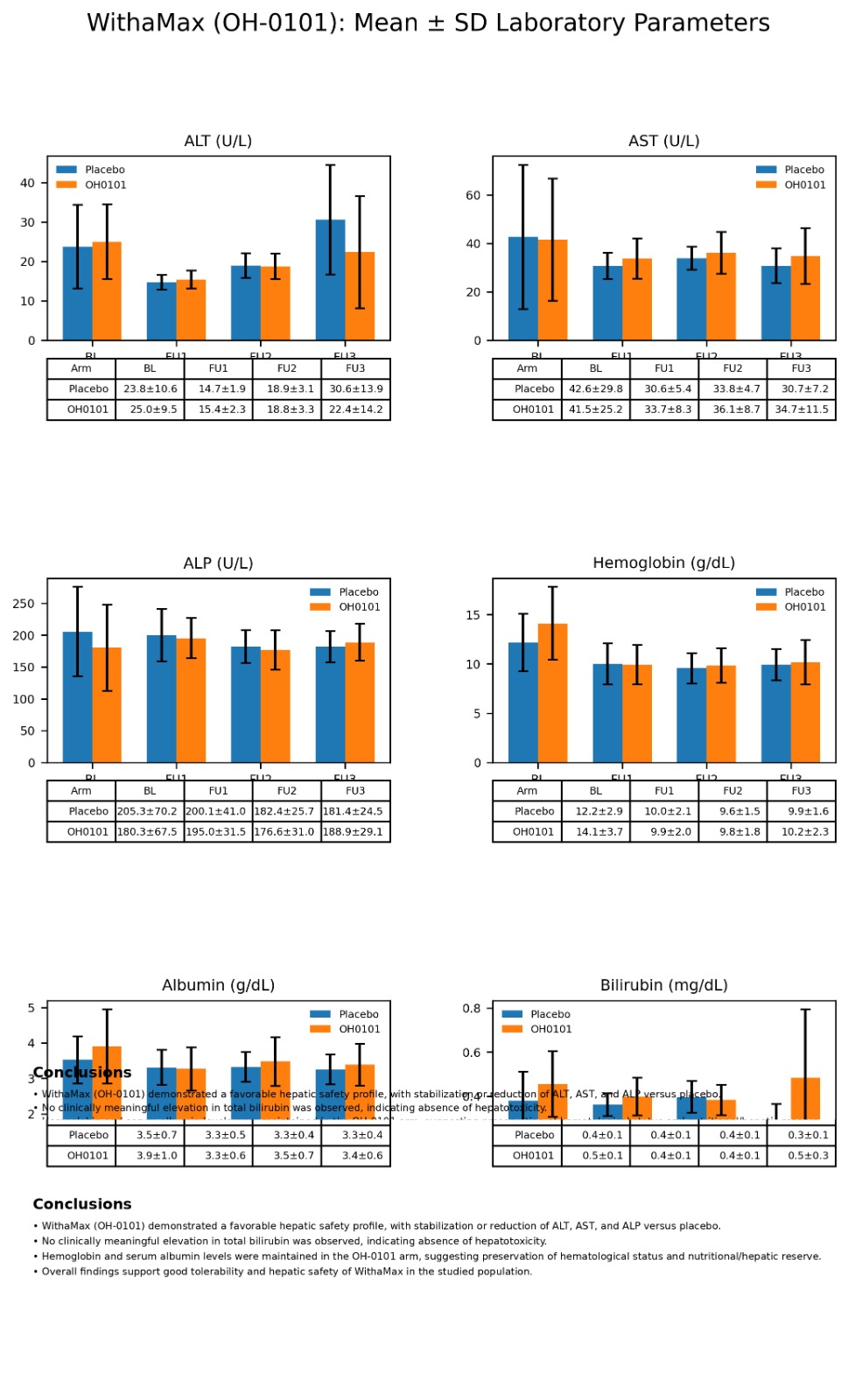
Safety evaluation included event-free survival analysis, physician global assessment, vital signs, and laboratory investigations. Laboratory parameters comprised liver function tests (ALT, AST, ALP, total bilirubin), hematological indices including hemoglobin, and serum albumin levels.
Efficacy evaluation focused primarily on quality-of-life outcomes measured using EORTC scoring systems. Functional scales (higher score indicating better functioning) and symptom scales (lower score indicating fewer symptoms) were analyzed. Statistical significance was assessed using Student’s t-test, with p < 0.05 considered significant.
Results
Across the eight-week treatment period, the investigational product demonstrated a favorable safety profile. Liver enzymes and bilirubin levels remained within clinically acceptable ranges in both arms. Stable or reduced ALT and ALP values were observed consistently in the investigational arm, with no clinically meaningful hepatotoxic signals. No serious adverse events were reported, and event-free survival was comparable between groups.
Hemoglobin levels were maintained throughout the study period, with marginal improvement trends in the investigational arm. Serum albumin values remained within normal limits in both groups, with slight preservation trends observed in subjects receiving OH-0101.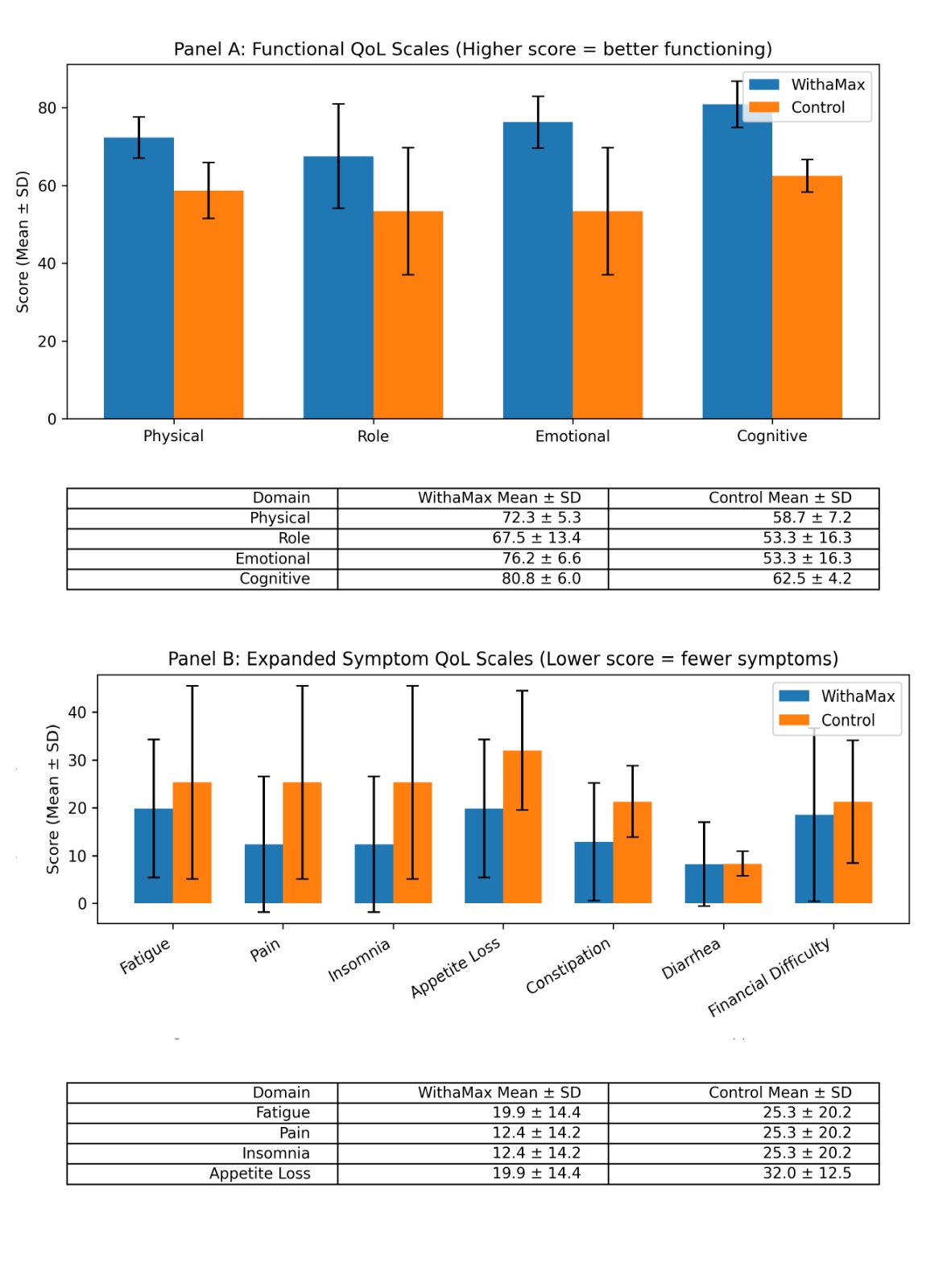
Quality-of-life assessment demonstrated statistically significant improvements in functional domains including physical, role, emotional, and cognitive functioning (p < 0.05). In symptom scales, reductions were observed in fatigue, pain, insomnia, and appetite loss. Overall symptom burden was lower in the investigational arm compared to control.
While laboratory parameter changes were clinically non-significant, overall trends favored the investigational arm without evidence of safety compromise.
Conclusions
In this proof-of-concept evaluation, OH-0101 was well tolerated in breast cancer patients undergoing chemotherapy and demonstrated statistically significant improvements in quality-of-life parameters. Hepatic and hematological markers remained stable, and no increase in adverse events was observed.
These findings support further investigation of adjunct supportive strategies aimed at improving systemic tolerance and patient-reported outcomes during active cancer treatment.
